Calculate pi of peptide chain8/11/2023  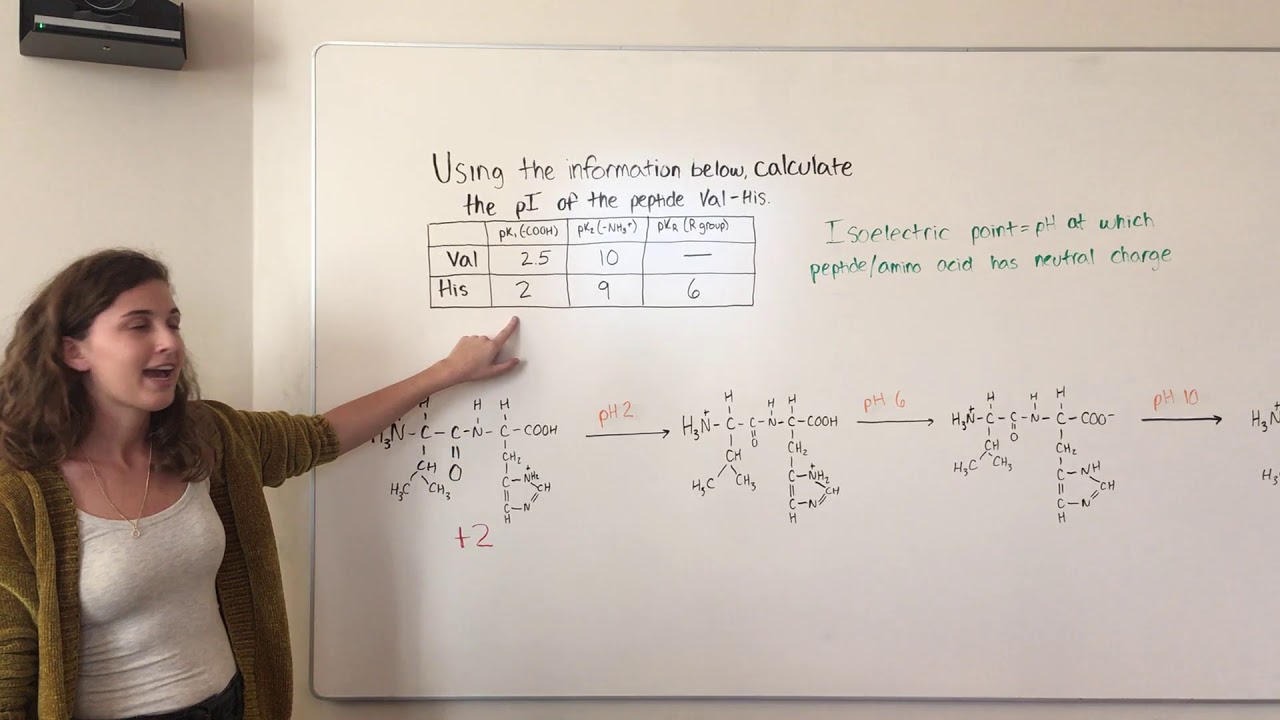
Above the highest pKa (10.53): All groups will be deprotonated, and the peptide will have a net charge of -1 (COO- and Glu side chain). 
We need to find the pH at which the net charge of the peptide is zero (isoelectric point, pI). Between the pKa values (2.34 - 10.53): The charges of the groups will change as the pH increases. Below the lowest pKa (2.34): All groups will be protonated, and the peptide will have a net charge of +2 (NH3+ and Lys side chain). It is widely used to steer design away from low solubility and aggregation and guide peptide separation and purification. Calculate the pI (isoelectric point) of a peptide chain by using the pKa values of the C-terminus, N-terminus, and R groups.DaChemOG ChemMadeSimple pI Is. The isoelectric point is the pH at which the average charge of the polyprotic acid is zero (0). The isoionic point or isoionic pH is obtained when a pure, neutral polyprotic acid is dissolved in water. Our pi prediction tool (11) is designed to calculate charge based on the side chains and carboxy- and amino-termini. We will consider three pH ranges: below the lowest pKa, between the pKa values, and above the highest pKa. The isoelectric point (pI) is a fundamental physicochemical property of peptides and proteins. Peptides and proteins are polyprotic systems which have an isoelectric or isoionic point (the pI ). Select an amino acid, then drag the pH arrow around to. Here are the acid-base equilibria for tyrosine: The form with no net charge is in red (+1 and -1 cancel out to give no net charge). False although there is no rotation about the peptide bond, the N-C and C-C. Since the p I is the p H at which the amino acid has no overall net charge, you need to average the p K a values relevant to the protonation/deprotonation of the form with no net charge. We will use the Henderson-Hasselbalch equation to calculate the charge of each ionizable group:Ĭharge = /( + ), where A- is the deprotonated form and HA is the protonated form.ģ. The pI is the pH at which the average charge of all of the amino acid species in solution is zero. To calculate the pI, first find the pH range where the zero charge state. 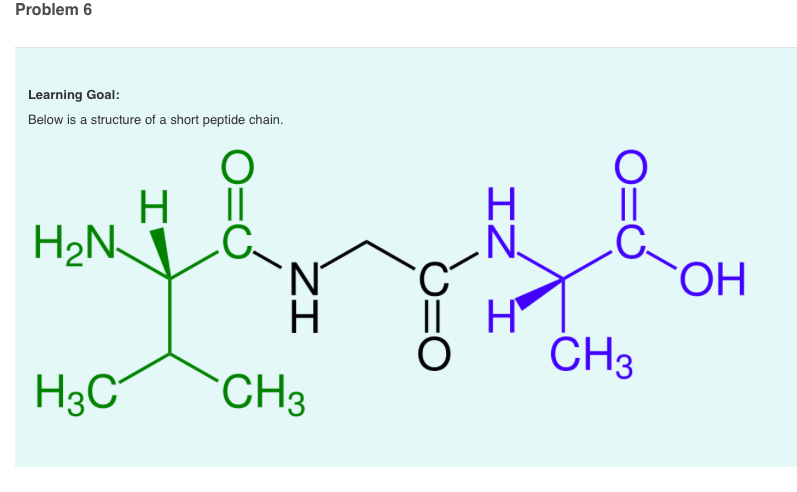
Now, we need to determine the charge of the peptide at different pH values. Lysine (Lys, K): side chain pKa = 10.53Ģ. Glycine (Gly, G): no ionizable side chain Our peptide calculator is a convenient tool for scientists as a molecular weight peptide calculator, which can be used as an amino acid calculator as well. Calculate the new pH value of the tripeptide solution (Relevant pKa values are 2.1, 4.2, 6.0. Glutamic acid (Glu, E): side chain pKa = 4.25 Beginning at pHpI you titrate 15mL of a 0.2M tetrapeptide. Alanine (Ala, A): no ionizable side chain The pKa values for the side chains and the N-terminal and C-terminal groups are as follows: First, we need to know the pKa values of the amino acids involved in the tetrapeptide. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
